Corrosion is estimated to be a $2.5 trillion problem globally. This revelation has led to implementation of several strategies by industry experts, organisation, regulatory bodies, researchers and many more. A visualisation of the concept of corrosion of materials may further enhance adoption of strategies. Which will result in improvements of corrosion resistance. Preventing corrosion may be classified into design and inspection; where several methods from design may be applied during the inspection stage of life of the pipe or structure. This phenomenon may be managed by: material selection, and retrofitting.
See also:
- Onshore Structural Design
- Compound/Complex Vibrating Equipment Foundation Design for Onshore Facilities
- Multi-Industry Piping and Pipeline Material Selection Decision Flowchart

A simplified collection of calculations for onshore structures can be obtained from here: https://www.amazon.com/ONSHORE-STRUCTURAL-DESIGN-STEP-CALCULATIONS/dp/B08Z3QPNP5
Also available in Nigeria for: ₦18,500.00 Contact – +2348182035528
Udemy course: Onshore Structural Design – Step by Step Calculations | Udemy
1.0 Material Design for Corrosion Prevention
1.1 Material Selection in view of Environmental Conditions
Material selection in view of environmental conditions should first be decided based on an organisations standards and approaches. In most cases, this approach complies with industry regulations. But for having an understanding as to how these organisational standards were developed in the first instance, and also to design for new cases, Figure 1 below is useful.
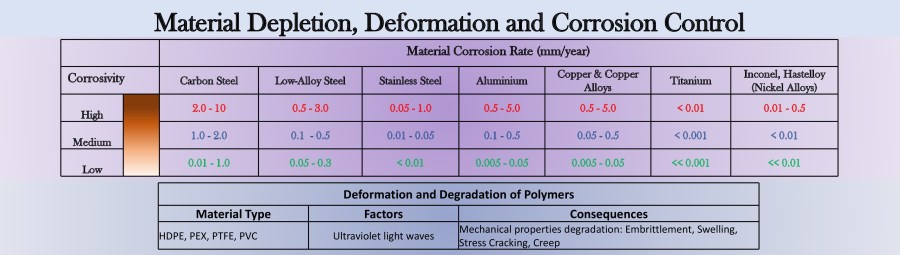
Corrosivity of the internal/external environment surrounding a structure or pipe determine the type material to be used. Additional criteria is the stress requirements and operating temperature. All these are in view of the cost and other lesser but important factors, such as applicability in the field. The corrosion estimates can be seen above which aids design decision for incorporating other corrosion prevention methods. Other reinforced plastic materials do not corrode; however, they are susceptible to ultraviolet, UV induced deformations.
Corrosion rates per material may be estimated mathematically. The extremes above for low, medium and high corrosivity for each material type is detailed above for thickness in millimeters per year depletion. More conservative corrosion depletion values may be estimated on a case by case basis that deviate from the conditions above.
1.2 Design Thickness of Pipe/Structure for Performance and Corrosion
Applying the relevant codes and standards to pipe/structure design, an additional thickness to cover for corrosion may be added. This additional thickness is as recommended by the code or based on the cumulative thickness depletion due to corrosion over the design life span of the structure. The formulas from these codes often consider the pressure, temperature and other stresses in determining the strength thickness, while corrosion thickness is a buffer.
1.3 Current Density of Metals and Corrosion Protection
Corrosion is an electrical process. Metals have a current density often measured in milli-Ampere/meter squared (mA/m2). This value is a measure of the speed of electron migration from these metals. Higher values indicate faster corrosion, while lower values indicate slower corrosion. This corrosion estimate aids in decision making regarding the method of corrosion protection to be employed. Here discussed are corrosion protection methods namely; coating, cathode protection and corrosion inhibitors.
1.3.1 Coating of Structures and Pipes
The purpose of a coating is to create a buffer/boundary distance between the environment and metal surface. Examples of coatings include: Fusion-Bonded Epoxy (FBE), Coal Tar Epoxy and Polyethylene Polypropylene (PE/PP). Before application of a coating, the corrosion rate or current density of carbon steel: 100 – 300mA/m2, stainless steel & Nickel Alloys: <5mA/m2. Upon application of coating materials, the current density reduces by 90-99%. For carbon steel + coating: 0.1 – 10 mA/m2, stainless steel + coating: <0.1 – <0.5 and Nickel Alloys + coating: <0.01 – <0.1mA/m2.
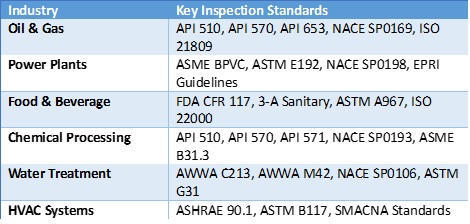
Coating thickness is determined by codes of practice and including the substrate material, operating temperature and design life span of the coating. Listed as follows are various coating types, their governing codes and example thickness. A summary of the various codes per industry is included at bottom of these specifications.
- FBE Coatings
- Governing standards:
- API RP 5L2 – Recommended practice for internal coatings of pipes.
- API RP 5L7 – External coatings for pipelines.
- NACE SP0394 – Application of FBE coatings.
- ISO 21809-2 – External FBE coatings for pipelines.
- AWWA C213 – FBE for water pipelines.
- Typical Thickness Requirements:
- Oil & Gas Pipelines: 250–500 µm (0.25mm-0.5mm or 10–20 mils).
- Water Pipelines: 300–500 µm (12–20 mils).
- High-temperature Applications: 400–800 µm (16–32 mils).
- Governing standards:
- Coal Tar Epoxy (CTE) Coatings
- Governing Standards:
- AWWA C210 – Liquid epoxy & coal tar for steel water pipes.
- API RP 5L2 – Internal coatings.
- NACE SP0188 – Coal tar epoxy for corrosion protection.
- ISO 15741 – Performance requirements for epoxy coatings.
- Typical Thickness Requirements:
- Marine & Offshore Structures: 500–1000 µm (20–40 mils).
- Buried Pipelines: 400–800 µm (16–32 mils).
- Water Pipelines: 400–600 µm (16–24 mils).
- Governing Standards:
- Polyethylene (PE) & Polypropylene (PP) Coatings
- Governing Standards:
- ISO 21809-1 – PE & PP coatings for pipelines.
- DIN 30670 – PE coatings for buried pipelines.
- CSA Z245.21 – Canadian standard for PE-coated pipes.
- AWWA C215 – PE coatings for water pipelines.
- Typical Thickness Requirements:
- 3-Layer PE/PP for Oil & Gas Pipelines:
- Total Thickness: 1.5–3.0 mm (60–120 mils).
- Adhesive Layer: 200–300 µm (8–12 mils).
- Top PE/PP Layer: 1.0–2.5 mm (40–100 mils).
- Water & Buried Pipelines: 1.0–2.5 mm (40–100 mils).
- 3-Layer PE/PP for Oil & Gas Pipelines:
- Governing Standards:
- Liquid Epoxy & Polyurethane Coatings
- Governing Standards:
- NACE SP0108 – Liquid epoxy coatings.
- AWWA C222 – Polyurethane coatings for water pipelines.
- ISO 12944 – Paint coatings for corrosion protection of steel.
- API 1169 – Pipeline construction and coating inspection.
- Typical Thickness Requirements:
- External Coating: 250–500 µm (10–20 mils).
- Internal Lining (Water Pipelines): 300–500 µm (12–20 mils).
- High-Performance Applications: Up to 1000 µm (40 mils).
- Governing Standards:
- Galvanizing (Zinc Coatings)
- Governing Standards:
- ASTM A123 – Hot-dip galvanizing for structural steel.
- ISO 1461 – Zinc coatings on iron & steel.
- BS EN ISO 14713 – Guidelines for galvanized coatings.
- AWWA C203 – Zinc coatings for water pipes.
- Typical Thickness Requirements:
- Structural Steel: 70–150 µm (2.8–6.0 mils).
- Pipelines: 100–200 µm (4.0–8.0 mils).
- Marine Applications: 150–300 µm (6.0–12.0 mils).
- Governing Standards:
- Thermal Spray Aluminum (TSA) & Thermal Spray Zinc (TSZ)
- Governing Standards:
- ISO 2063 – TSA and TSZ coatings for corrosion protection.
- NACE SP0187 – Thermal spray coatings for pipelines.
- API RP 2F – Offshore structures coating requirements.
- Typical Thickness Requirements:
- TSA for Offshore Pipelines & Structures: 150–300 µm (6–12 mils).
- TSZ for Industrial Equipment: 100–250 µm (4–10 mils).
- Governing Standards:
1.3.2 Cathode Protection
Cathode protection is a balancing of the electrochemical reaction occurring between a structure and the environment by substituting electron loss through a different source other than the metal structure or pipe. There exists Sacrificial Anode CP (SACP) and Impressed Current CP (ICCP). SACP utilises the natural galvanic potential of a metal to supply electrons for the process. For example, Aluminium may be used as a sacrificial anode seeing its high natural potential for migration of electrons. For ICCP, an external current is used to activate the reaction. In this case, materials which are less reactive such as graphite may be used to provide the current protection. Below is a guide on how to estimate the current for both SACP, ICCP and to quantify the mass of sacrificial anode to be used.
1.3.3 Corrosion Inhibitors
Corrosion inhibitors are chemical compounds that slow down or prevent corrosion when added to a corrosive environment. They work by forming a protective film on the metal surface or altering the corrosive medium’s properties. These are introduced into pipes and storage tanks in order to react with the surface of the metals and create a protection.
2.0 Inspection and Control of Corrosion
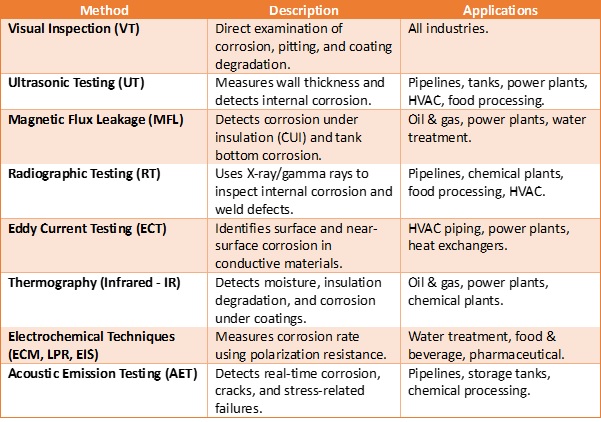
In the operating phase of facilities, inspection of installations takes center stage. From ascertaining the effectiveness of design assumptions to response of accessories to corrosion effects. A desk study is a necessary step for an overarching strategy for corrosion inspection and assessment. From this preliminary study; facility register, location, layouts of facilities, material types, age and previous corrosion protection an understanding of the situation in the field can be quickly understood prior to site visit. There are several non-destructive tests (NDTs) to check for thickness variations, and electrochemical effects such as in the use of reference electrodes to evaluate the state of corrosion of the pipe or structure.
2.1 NDT for Inspecting Corrosion and Governing Codes
Corrosion inspection in various industries follows specific non-destructive testing (NDT) techniques and codes/standards to ensure structural integrity, safety, and compliance.
2.2 Inspection of Cathode Protection Utilising Reference Electrodes
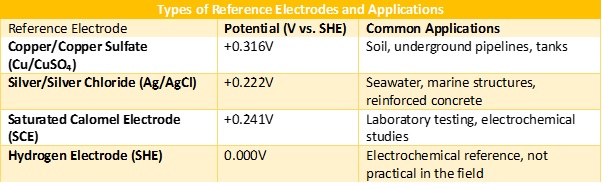
A reference electrode is a stable electrode with a known, constant potential, used to measure and monitor the electrochemical potential of a metal structure in corrosion control applications. It serves as a reference point for comparing the electrical potential of the metal being inspected. Listed below are the functions of reference electrode system for inspecting cathode protection, types of reference electrodes and codes of practice.
2.2.1 Key Functions of a Reference Electrode in Corrosion Inspection
- Measuring Corrosion Potential (Half-Cell Potential)
- Used in Cathodic Protection (CP) systems to measure the potential of a protected metal structure (e.g., pipelines, tanks, offshore platforms).
- Helps determine if a metal is in an active or passive corrosion state.
- Example: Carbon steel pipelines should maintain a potential below -850 mV (vs. Cu/CuSO₄ electrode) in soil.
- Verifying Cathodic Protection (CP) Effectiveness
- Ensures that impressed current or sacrificial anodes provide sufficient protection.
- Prevents overprotection, which can cause hydrogen embrittlement in high-strength steels.
- Used in onshore pipelines, marine structures, and storage tanks.
- Detecting Coating Defects & Corrosion Hotspots
- Used in DCVG (Direct Current Voltage Gradient) and CIPS (Close Interval Potential Survey) techniques to locate coating defects on buried pipelines.
- Identifies areas of active corrosion under coatings.
- Assessing Corrosion Rate in Electrochemical Testing
- In Linear Polarization Resistance (LPR), Electrochemical Impedance Spectroscopy (EIS), and Potentiodynamic Testing, reference electrodes measure the corrosion rate of metals in solutions.
- Used in laboratories and field corrosion studies.
- Monitoring Corrosion in Reinforced Concrete
- Reference electrodes (e.g., Silver/Silver Chloride) are embedded in concrete to measure rebar potential.
- Helps prevent chloride-induced corrosion in bridges and buildings.
- Example: Pipeline Corrosion Potential Measurement Using Cu/CuSO₄
- Place the Cu/CuSO₄ reference electrode in contact with the soil near the pipeline.
- Connect a voltmeter between the pipeline and the reference electrode.
- Measure the pipe-to-soil potential.
- If < -850 mV → Proper cathodic protection.
- If > -850 mV → Inadequate protection, corrosion risk.
- If < -1200 mV → Overprotection risk (hydrogen embrittlement).
2.2.2 Codes of Practice and Standards Governing Reference Electrodes for Corrosion Inspection
Several international codes and standards define the use, installation, and calibration of reference electrodes in corrosion inspection, cathodic protection (CP), and electrochemical testing. These standards ensure accurate corrosion potential measurements for pipelines, tanks, offshore structures, and reinforced concrete.
- General Standards for Reference Electrodes in Corrosion Inspection
- Cathodic Protection (CP) Standards Using Reference Electrodes
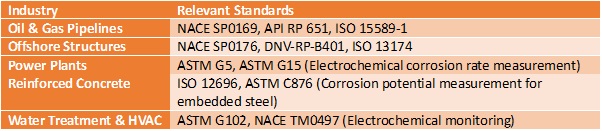
- Reference Electrodes for Corrosion Monitoring in Specific Industries
- Reference Electrode Calibration and Testing Standards
2.3 Corrosion Control Measures
Upon ascertaining the condition of pipes and structures, a decision may be made as to the course of action. Where it has proven necessary, the principles applied in the design of materials and prevention are again applied in control. In this instance of control, practicability of a solution is paramount.
3.0 Further Readings:
- Corrosion: Corrosion Protection for the Oil & Gas Industry Pipelines, Subsea Equipment and Structures Author: Mavis Sika Okyere
- Coating & Painting: Essentials of Coating, Painting, Lining for the Oil, Gas and Petrochemical Industries Author: Alireza Bahadori






slick! Global Tea Industry Adopts Sustainable Practices 2025 cute